Moving towards 80,000 Hours of Fuel Cell Operation and Beyond
Novel and innovative ways of maximising product utilisation and minimising product footprint are crucial in establishing a true alternative to conventional solutions.
Hydrogen-fuelled low temperature polymer electrolyte fuel cells have throughout the years been regarded as a viable and sustainable option in the transition to a green energy supply chain from a technological point of view, yet a lack of pressure in often purely economically driven trade-offs have for a long time hindered a large scale roll-out. As the impact and the accompanying challenges posed by climate change are becoming increasingly pressing and a threat to life as we know it, hydrogen as an energy carrier and fuel cells as a highly efficient converter thereof, are experiencing unprecedented attention.
As a price tag is more and more often put on the emission of pollutants and greenhouse gases, economic viability shifts too. However, this abruptly increased interest in the technology has put an industry, which is in the critical phase of the transition from a small scale and research-driven mind-set to mass manufacturing and the holistic analysis of processes in the spotlight. Important questions in this transition process largely concern quality assurance, reliability, predictability and extension of the lifetime of fuel cell systems as well as the inclusion of the entire manufacturing and utilisation chain in a circular economy. Economically, fuel cells are as of now still in a disadvantaged position in comparison to more established technologies, such as the internal combustion engine. However, several means to close this gap are in reach.
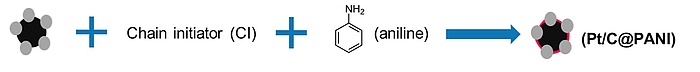
Figure 2: Fuel cell catalyst synthesis.

Figure 2b: TEM image of the deposited protective layer on the fuel cell catalyst.
Connecting the Dots
Beyond the obvious economy of scale, which is anticipated to come into effect as the industry shifts towards mass manufacturing, improving material utilisation – in particular of the high-cost, mostly platinum-based electrocatalyst – is a key factor in cost reduction. Research includes the development of new catalysts with a higher activity, paired with a lower Pt loading, as well as the stabilisation of catalyst and support.
Another critical electrochemically active component is the polymer electrolyte, which is needed both as the membrane and as a part of the electrodes to provide ionic conductivity. As the membrane separates both reactants, the chemical and physical stability are crucial not only to performance, efficiency and lifetime, but also to safety.
Both polymer electrolyte membrane and catalyst exhibit a large interconnection between their chemical composition, physical properties and their performance and durability. The Institute of Chemical Engineering and Environmental Technologies has a long history in the investigation of degradation mechanisms of fuel cells and their prevention as well as in the development of materials for the use in fuel cells. Through the increasing understanding of degradation mechanisms, their connection to the used materials and their impact on the behaviour of fuel cells, increasingly advanced monitoring techniques emerge, which are crucial to long term reliable fuel cell operation.

Figure 3:CEET staff working in the fuel cell laboratory.
Reducing the Footprint of Fuel Cells
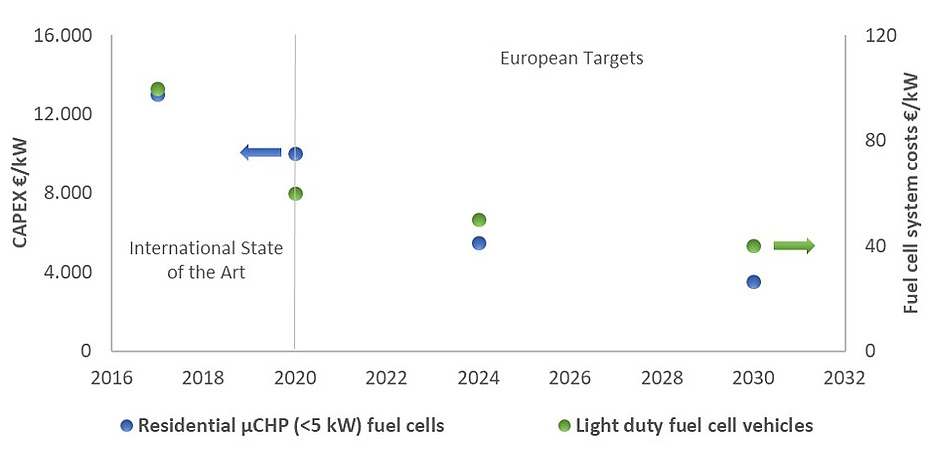
Every technology possesses an individual footprint, be it based on renewable resources or not. This naturally includes fuel cells and as production is rapidly upscaling, these processes are experiencing increasing attention as well. Research and development is aiming at the implementation of new and resource-efficient manufacturing techniques. Another path towards a smaller footprint, however, is to increase the duration of the fuel cell utilisation. Targets set for fuel cells, such as the one of 80,000 h for stationary applications given by the US Department of Energy [1], are, however, difficult to prove in a reasonable time, as this is equivalent to 10 years of up-time. Relying entirely on use case data would result in unacceptably long development iteration intervals. It is furthermore worth noting that in these orders of magnitude, the contribution of system components becomes critical as well and the actual duration of the fuel cell may be overshadowed by degradation of other components, further complicating the proof of such lifetime durations. One of the longest running systems reported has exceeded 65,000 h of operation within 12 years [2] and is still counting.
Therefore, in order to accelerate the development process, accelerated stress tests are made use of. Individual stressors are selected based on the respective use case or targeted material and deliberately applied in an operation profile. This allows for a far quicker identification of future lifetime limitations of the fuel cell itself.
With this information, it is possible to give lifetime estimations in a reasonable time. At the same time, triggering specific degradation mechanisms allows us to understand the impact they have on the stability and behaviour of the fuel cell. This behaviour can then – like a fingerprint – be looked for in use case data, giving an early insight into ongoing degradation and allowing fast actions for mitigation.
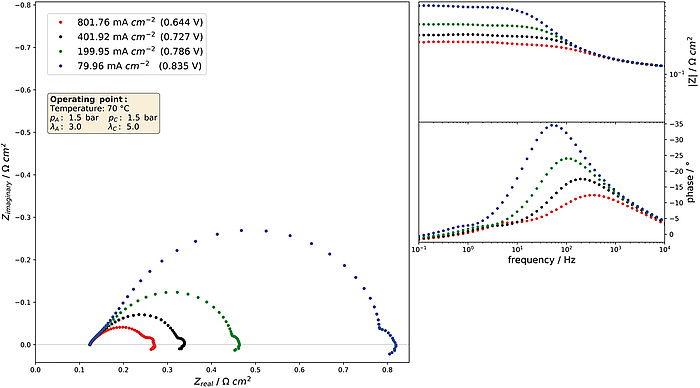
Figure 5: Electrochemical impedance spectra at differnt operating points.
References:
- Durability working group, Hydrogen and Fuel Cell Technologies Office, U.S. Department of Energy; www.energy.gov/eere/fuelcells/durability-working-group (accessed 23/08/2021)
- Nedstack fuel cell technology: 65.000 running hours and counting; nedstack.com/en/ news/65000-running-hours-and-counting (accessed 20/08/2021)
Kontakt
Merit Bodner has been working in the field of fuel cell research since 2011, with particular focus on degradation phenomena and lifetime limitations of low-temperature polymer electrolyte fuel cells. After her graduation, she had leading positions at Danish Power Systems in Denmark and Nedstack Fuel Cell Technology in the Netherlands. Since May 2021, she has held a tenure-track position at Graz University of Technology. Merit Bodner has over 40 scientific publications and presentations and has contributed scientifically as an expert in commissions and in the peer-reviewing process.



