Abbas Group . Supercapacitors . Fast Energy Storage (Coupling of Battery & EDL Electrodes)

Materials.
Nanoporous carbons: Our group uses high surface area carbons (1500-2500 m2/g) as electrodes which possess majority of pores in the range of 1 nm making them ideal for charge storage. The electronic conductivity of these carbons is fairly high which assists in cycling at very high rates (time constant, τ < 1s). The surface functional groups on carbon electrodes generated during long-term charge/discharge cause performance decay. We implement various strategies from porosity tuning to pretreatment of electrodes in order to maintain their physicochemical and electrochemical stability.
Eco-friendly electrolytes: Up to 35 wt.% of a supercapacitor consists of electrolyte. We use environmentally friendly and green electrolytes from diluted aqueous electrolytes to water-in-salt electrolytes as well as deep eutectic solutions, all from low-cost chemicals. The selection rule for electrolyte is high ionic conductivity, low viscosity and excellent dielectric properties.
Systems.
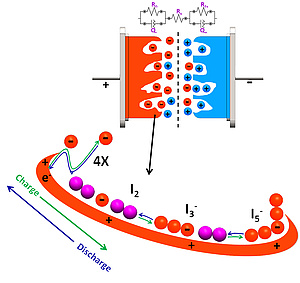
Hybrid supercapacitors: Hybrid supercapacitors are used where high power is required, such as uninterruptible power supply (UPS), automotive hybridizations and backup applications. In hybrid supercapacitors, one electrode is made to performance like a battery while keeping the other electrode as electric double-layer (EDL) charge storage. This helps to enhance the energy density. Our group takes advantage of highly reversible iodine electrochemistry to in situ generate a battery electrode from nanoporous carbon materials. The high surface area and narrow pore size distribution helps to store large quantities of iodine in the nanopores making it a high capacity electrode, and overall high energy density hybrid SCs.
Electric double-layer capacitors (EDLCs): In an EDLC, both positive and negative electrodes store charges at the EDL. Since the charge storage is physical, the output power in EDLCs is quite high in the range 5-10 kW/kg. We use highly stable ionic liquids as electrolytes to produce EDLCs and test their life cycle under harsh climatic conditions (+60°C to -40°C).

Technology.
Prototypes: To produce pouch cells, the carbon material in homogenous slurry form is applied to the current collector (Al or stainless steel) foil. The electrode is calendared after preliminary drying and then moisture is removed by extensive drying at 100°C for several hours. The coated electrodes are coupled together to produce pouch cells in the size of 10 x 10 cm2. Our work stations can test both single and stacked pouch cells up to a current range of 10 A.

Dr. techn. habil. Qamar Abbas
Institute for Chemistry and Technology of Materials
Stremayrgasse 9
A-8010 Graz
Phone: +43 (0)316 873-32357
qamar.abbas@tugraz.at
Projects:
FWF funded List-Meitner Fellowship 2019-2021
EPU ERA-NET 2020-2021
FFG funded ECOCAPS 2021-2025