News

 Is there carbenoid activity in transesterification reactions catalyzed by azole/oxirane pairs?
Is there carbenoid activity in transesterification reactions catalyzed by azole/oxirane pairs?
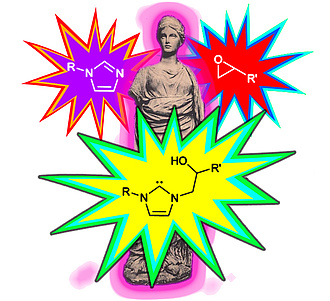
Building on our previous work on carbenoid reactivity from azoles and oxiranes, we studied its importance in transesterification reactions. Various combinations of commercially available Lewis bases (1-methylimidazole, 4-(dimethylamino)pyridine, 1,4-diazabicyclo[2.2.2]octan and 1,8-diazabicyclo[5.4.0]undec-7-ene) were employed alongside phenyl glycidyl ether as a co-catalyst for the polymerization and depolymerization of polyester compounds. During glycolysis-based degradation of commercial-grade poly(ethylene terephthalate), we found that combining 1-methylimidazole with oxirane drastically reduced degradation time. This phenomenon can be explained by the formation of an N-heterocyclic carbene in situ through the reaction between the azole and the oxirane. In the polymerization of ε-caprolactone, all Lewis bases showed enhanced activity when used with oxirane. Here, the stationary concentration of alkoxides is assumed to determine the speed of ε-caprolactone polymerization, which is fasted when the formulations contain a Lewis base, the oxirane, and an alcohol initiator.
 Easy access to carbenoid activity!
Easy access to carbenoid activity!
In a recent work published in Advanced Synthesis and Catalysis, researchers from the Christian Doppler Laboratory of Organocatalysis in Polymerization disclosed a modular and potentially inexpensive method of generating carbenoid reactivity in situ. Various combinations of azole compounds and oxiranes have been shown to catalyze benzoin condensation at elevated temperatures. Oxygen exclusion is unnecessary under the chosen reaction conditions.
They demonstrated the practical utility of this catalytic system by polymerizing simple, bifunctional, aldehyde/oxirane-containing monomers (e.g., the glycidylether of vanillin) using 5 mol% 1-methylimidazole in a solventless manner and without excluding air. The monomers polymerized via the formyl and oxirane groups and yielded thermoset materials with glass transition temperatures above 100 °C.
 From 1D to 3D: How NMR can resolve a sequence of Li diffusion pathways
From 1D to 3D: How NMR can resolve a sequence of Li diffusion pathways
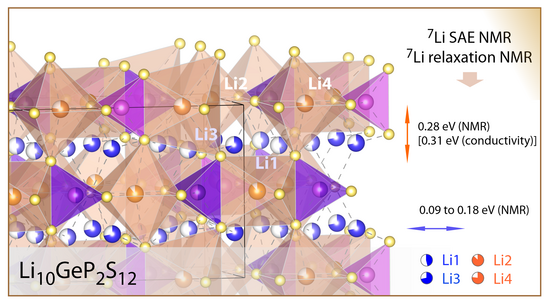
We used 7Li spin-alignment echo and complementary NMR techniques to track Li-ion motion in Li10GeP2S12 across nearly 12 orders of magnitude in jump rates, from ultraslow local exchange at 90 K to rapid long-range diffusion near room temperature. The data reveal a clear transition from 1D/2D to 3D Li+ transport with distinct activation regimes. This provides a detailed picture of how dimensionality governs ion dynamics in one of the fastest solid electrolytes known. The JACS paper available online.
 Revealing formation reactions of lead-free BCZT piezoceramics
Revealing formation reactions of lead-free BCZT piezoceramics
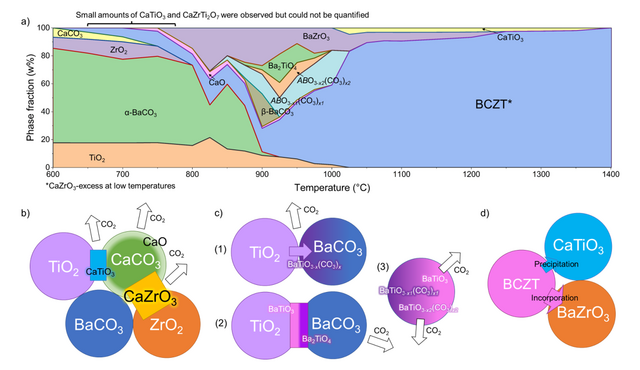
Continuous effort is focused on improving lead-free piezoelectric materials, including the piezoceramic (Ba,Ca)(Zr,Ti)O3. However, unwanted secondary phases may have unpredictable influence on the performance and decrease the reproducibility. In our latest study, we used in-situ X-ray diffraction and thermal analysis to investigate the solid-state phase formation sequence of (Ba,Ca)(Zr,Ti)O3. Based on the identified formation conditions of secondary phases (see figure below), process adaptations could be applied to not only increase the phase purity, but also enhance the piezoelectric performance. Unexpectedly, we also found a new intermediate phase that could change the view on the reaction mechanism. Read more in our recent publication in the Journal of the European Ceramic Society.
10th EuChemS Chemistry Congress
July 12-16, 2026
Antwerp, Belgium
YPRA 2026
June 29 - July 1, 2026
Leoben, Austria
12th Conference on Green Chemistry & Nanotechnologies in Polymeric Materials
June 15-17, 2026
San Sebastian, Spain
notabene: Twitter / X is still used by some of us as a news medium (for science), but we explicitly distance ourselves from the partly crude conspiracy theories and unverifiable "facts" that are often published.
You may also follow some of us on LinkedIn: