Chemical recycling of natural rubber
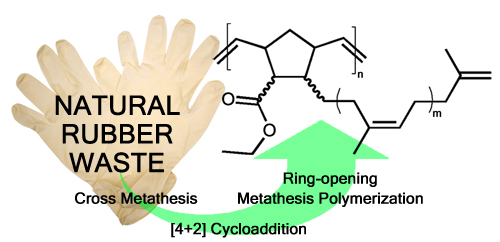
Natural rubber is the most widely used feedstock in elastomer production owing the unique properties of this agricultural product. Natural rubber production amounted to more than 13 million metric tons in 2017 and is used in countless applications spread across many important industries, such as chemical, transportation or medical industries. The end-of-life treatment of such crosslinked thermosets is typically incineration. However, efforts to chemically recycle natural rubber products have received ever increasing importance. Cross metathesis with ethene (ethenolysis) or other alkenes is thought to be a promising way to convert rubber waste into feedstock for the chemical industry.
Yet researchers of ICTM reported their results on using electron deficient olefins, namely acrylates, as the cross metathesis partner in the degradation of natural rubber waste in form of gloves frequently used in the laboratory.
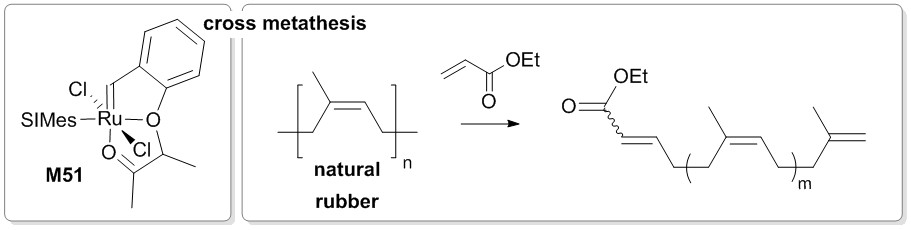
An efficient degradation protocol was elaborated which allows for the scalable synthesis of enoate-end capped oligo-cis-isoprene (typically m = 3). The created enoate-group now offers entry to a manifold of further transformations of the rubber degradation product. The said versatility is demonstrated by introducing a norbornene group to the oligo-cis-isoprenes rendering them polymerizable. Polymerisation via Ring-opening Metathesis Polymerization yielded a main- and sidechain unsaturated polymer featuring a glass transition temperature of about -60°C (and a thermal stability up to 270 °C), which now again qualifies as component in vulcanizable compounds.
For further reading see the open-access article:

"
Converting natural rubber waste into Ring-opening Metathesis polymers with oligo-1,4-cis-isoprene sidechains."
Abbas, M.; Neubauer, M.; Slugovc, C.
Polym. Chem. 2018,
9, 1763-1766.
DOI: 10.1039/C8PY00233A
The work was featured on
All Things Metathesis