Analytical Techniques for the Identification and Quantification of Odor Active Substances
The analysis of odor active substances seems to be quite an easy task because odor activity can only occur if a molecule is volatile and the molecular weight is below 400 Dalton. So the “gold” standard for analysis is high resolution gas chromatography which is one of the most advanced and efficient separation tools.
But the limiting factor is the human nose which is able to perceive odor activity over 10 to 12 orders of magnitude reaching sometimes incredibly low sensory thresholds as given in table 1. In addition, only up to 3% of the volatile substances show odor activity, so the other way round you face 97% of possible interfering compounds.
*T. Hoffmann et al., Angew. Chem. Int. Ed. 2014, 53, 7124 – 7143
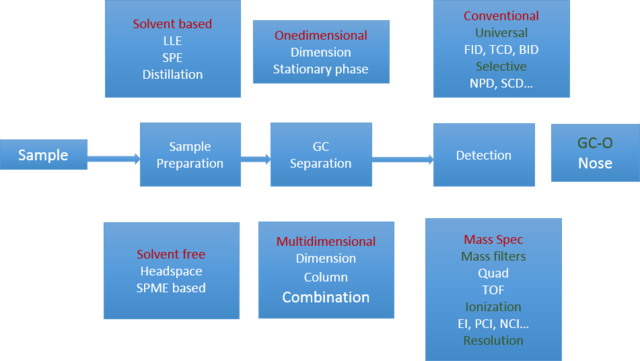
**F. B. Whitfield et al., J. Agric. Food Chem., 1997, 45 (3), 889–893 So to handle these problems there is a strong need for SENSITIVE and SELECTIVE analytical methods which consist of sampling, sample preparation, separation and detection (Figure 2)
| Substance | Descriptor | Sensory Threshold* [µg/L] in water | |
|---|---|---|---|
| ethanol | alcoholic | 990.000 | |
| 2-methyl-1-propanol | malty | 19.000 | |
| acetic acid | acid, vinegar | 5.600 | |
| 1-hexanol | green, grassy | 590 | |
| 2-phenylethanol | flowery, wine | 18 | |
| 2-methoxy-4-vinylphenol | smoky | 5 | |
| butan-2,3-dione | butter, fermented milk | 1 | |
| 3-(methylthio)propanal | cooked potato | 0.4 | |
| 2-acetyl-1-pyrroline | popcorn | 0.05 | |
| (E,E)-2,4-decadienal | fatty, french fries | 0.03 | |
| wine lactone | coconut | 0.02 | |
| (E)-β-damascenone | cooked apple | 0.01 | |
| (E,Z)-2,6-nonadienal | cucumber | 0.005 | |
| 1-p-menthene-8-thiol | grapefruit | 0.000 2 | |
| 2-methyl-3-furanthiol | meaty, bouillon | 0.000 03 | |
| 2,4,6-tribromoanisole** | musty, mouldy | 0.000 02 |
*T. Hoffmann et al., Angew. Chem. Int. Ed. 2014, 53, 7124 – 7143
**F. B. Whitfield et al., J. Agric. Food Chem., 1997, 45 (3), 889–893 So to handle these problems there is a strong need for SENSITIVE and SELECTIVE analytical methods which consist of sampling, sample preparation, separation and detection (Figure 2)