Silicates/Oxides formation and weathering
Understanding the important role of clay minerals – the by far most abundant silicates on the Earth`s surface –as (trace) element sources and sinks in modern and past sedimentary and marine-diagenetic environments provides substantial information about fluid-rock interactions, paleoclimate, global element cycling, and fundamental biogeochemical processes at water-mineral interfaces.
The experimental synthesis and/or dissolution of redox- and climate-sensitive clay minerals such as glauconite, interstratified illite-smectite, Fe-Mg-saponite, sepiolite and (amorphous) aluminosilicates are powerful tools to trace the underlying reaction pathways and kinetics of clay mineral reactions at low temperatures. Herein, we use latest-state-of-the-art solid-phase and fluid-phase characterization methods as well as hydrogeochemical modeling in order to link our novel experimental work with processes occurring in our dynamic system Earth.
Specific tasks can be seen in the following sections.
Sr and Mg isotope fractionation during basalt alteration
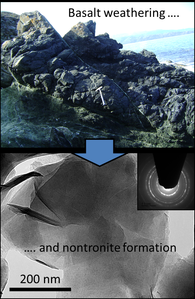
Neo-formed oceanic crust (e.g. basalt) in contact with seawater undergoes various forms of chemical alteration, typically leading to the formation of clay minerals and sulfate minerals (see SEM image of altered crystalline basalt below). In this project, we investigate the fate of critical trace elements and isotopes of Sr and Mg during basalt-seawater interaction in order to shed light on the processes and physicochemical parameters that control the (re)cycling of elements at hydrothermal spots of the deep ocean (e.g. mid-oceanic ridge sites).
Contact: Andre Baldermann
(Ad)sorption properties of short-rage-order aluminosilicates
Pollution of water by (heavy) metal ions is a major environmental issue worldwide, causing deleterious effects on human health, aquatic life and to the terrestrial environment. Thus, there is an increasing demand for the development of novel natural and synthetic adsorbents that can be used for water clean-up.
The use of clay minerals for wastewater treatment can be advantageous over other recently available adsorbents due to their worldwide occurrence, low costs and unique surface (charge) and physicochemical properties. Herein, we investigate the potential of natural and synthetic short-rage-order aluminosilicates (allophane and imogolite) for remediating wastewater that is severely contaminated with hazardous metal ions.
Contact: Andre Baldermann
Clay mineral proxies in paleo-climate studies
The mineralogical, structural and chemical signatures of authigenic and detrital clay minerals can be successfully used to reconstruct the provenance, depositional history and the climatic evolution of terrestrial basins and marine sedimentary successions throughout the geological record.
In this project, we study the sedimentology, mineralogy, pollen record, major and trace element composition and isotopic (e.g. δ13C, δ18O) composition of mostly siliciclastic and carbonate-bearing sediments from several key locations worldwide, such as the highly fossiliferous deposits from the Valley of Lakes (Mongolia) and the oil shales from the Caspian Sea (Azerbaijan) in order to shed light on the paleo-climatic evolution of these hot spots during the Cenozoic.
Contact: Andre Baldermann
Glauconite
Green-clay authigenesis is a worldwide occurring phenomenon, covering huge areas of shelf and ocean floor sediments. We investigate the environmental conditions and reaction paths of the so-called Fe-smectite to glauconite reaction in shallow- and deep-water settings during early diagenesis using a multi-method approach that comprises various solid-phase and fluid-phase analysis as well as hydrogeochemical and kinetic modeling.
The quantification of ultimate iron sequestration related to green-clay authigenesis in modern, deep sea environments is a major task of our ongoing research affecting a variety of biogeochemical processes in the low-temperature cycle of iron.
Contact: Andre Baldermann
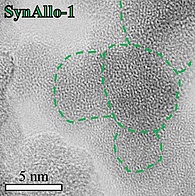
Experimental synthesis of ferruginous clay minerals
Ferrous saponite and nontronite are the major alteration products of submarine basalt formed at mid-oceanic ridge sites. The precipitation and deposition of ferruginous clay minerals on the ocean floor is well-known to limit the supply of dissolved iron to the overlying oceanic water masses, which affects a variety of biogeochemical processes in deep-sea sediments and in the deep biosphere environment in general. Thus, an improved understanding of the ambient environmental controls affecting the distribution and abundance of authigenic ferruginous clay minerals in the sedimentary record may provide substantial information about the processes operating the modern and past biogeochemical cycle of iron.
In this project, we synthesize ferrous saponite and nontronite in Teflon-lined, stainless steel autoclaves at diagenetic temperatures, alkaline pH, reducing conditions, and using experimental solutions with varying molar Si:Fe:Mg ratios in order to investigate the effect of these key parameters on the mineralogy, structure, stability, and chemical composition of laboratory grown ferrous clay minerals. Measurements of stable Fe, Si, and Mg isotopes are planned to resolve the underlying reaction mechanisms linked to the formation of saponite and nontronite.
Contact: Andre Baldermann
Experimental synthesis of sepiolite
The formation of Mg-silicates and modulated Mg-phyllosilicates at Earth`s surface conditions has been used successfully for the reconstruction of sedimentary facies in modern and ancient lacustrine, peri-marine, and diagenetic environments. To our current knowledge, sepiolite – a hydrous Mg-rich clay mineral with a modulated structure – is the only Mg-silicate which forms by direct precipitation from seawater.
Using a novel experimental approach, we precipitate sepiolite under nature-near conditions at ambient temperatures in order to experimentally derive the apparent growth rates of fibrous sepiolite crystals. Mg and Si isotope analysis of synthetic and naturally occurring sepiolite is used to trace the underlying mineral-forming processes that are associated with the co-precipitation of Mg2+ ions and silica.
Contact: Andre Baldermann
Chert formation and proxy development (Si/Al, Si-isotopes)
The deposition of widespread phosphorites at the Precambrian-Cambrian boundary is often associated with the deposition of abundant bedded chert sequences in shallow and deeper water environments. However, so far, neither the source of silica nor the formational mechanisms are known with certainty.
We therefore investigate how the remarkable retrieval and concentration of dissolved Si can be achieved, how weathering, hydrothermal activity and/or bio-engineering influenced the oceanic and depositional system. This is achieved by applying state-of –the-art geochemical methods, such as trace element ratios, REE, light stable (Δ17O) and Si isotopes.
Contact: Dorothee Hippler