Carbonate in natural and man-made surroundings
Specific tasks: Dolomite
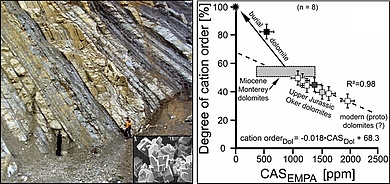
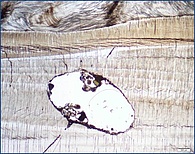
Dolomite, CaMg(CO3)2, is a common rock-forming mineral in ancient platform carbonates, but its occurrence in modern environments is rare, despite the ambient seawater is highly supersaturated in respect to this mineral phase. After more than 200 years of ongoing research there is still an active debate in the geoscientific community about the underlying processes causing dolomite formation in sedimentary (low temperature) settings. This enigma is referred to as the dolomite problem.
In this sub-project, we investigate spatial trace element distributions and concentrations (e.g., Na, Sr, S, Fe, Mn), the isotopic signatures (e.g., δ13C, δ18O, δ26Mg, and δ34SCAS) as well as the petrographic and mineralogical features of recent dolomites and ancient dolomitized platform carbonates in order to elucidate the physicochemical conditions and underlying dolomitization pathways operating in modern and past dolomite-forming environments.
Contact: Andre Baldermann
Non-traditional stable isotope fractionation of lacustrine high-Mg calcites and dolomite of Lake Neusiedl
Lacustrine carbonates display an important regional environmental archive, with deposition depending upon lake hydrology and morphology, and hence climatic influences. It is further known that carbonate deposition in lakes is mainly driven by biogenic or bio-induced precipitation, resulting in some places in the enigmatic formation of high-Mg calcite or dolomite.
Lakes, such as Lake Neusiedl are thus ideal large-scale laboratories that permit detailed examination of highly important aspects of authigenic dolomite formation. Calcium and magnesium isotope signatures trace Ca and Mg cycling within the lake and its catchment area in relation to authigenic high-Mg calcite and dolomite formation.
Contact: Dorothee Hippler
Biomineralisation vs. Bioerosion-induced alteration of biogenic carbonates (CHARON)
The shells of marine calcifiers (e.g. bivalves) are mainly composite materials made of mineralized and organic tissue. This fact makes shell material highly interesting for hard substrate dwellers, such as micro-boring algae or fungi, causing remarkable bioerosion of the original shell material. This study therefore focuses on the impact of bioerosion on the structure, mineralogy and geochemistry of original shell material via the comparision of pristine, bio-eroded and fossil shell material.
Contact: Dorothee Hippler
Mineral precipitates in geotechnical drainage systems
Rapid deposition of chemical sediments (mostly carbonates and sulfates) is widespread in man-made drainage settings such as artificial (bypassing) river beds and tunnel drainage systems. This constitutes a serious issue if deposition and clogging (scaling) affects the water transport in water pipe systems. Pronounced fluid-solid interaction results in dissolution versus precipitation reactions, e.g. concrete linings interacting with stream waters promoting supersaturated aqueous solutions.
Our research focuses is on hydrogeochemical and site-specific process understanding with regard to applicable environmental and engineering solutions. In order to decipher the distinct scale formation mechanisms and kinetics, we investigate in particular the nucleation and growth dynamics of carbonate minerals. This approach comprises detailed petrographic analysis, using high-resolution techniques (e.g. scanning electron microscopy) and also mineral and water characterization combined with in-situ monitoring programs and hydrochemical modelling.
Contact: Ronny Boch
Monitoring and Assessment of Nucleation and Growth of Carbonates in Water Pipe Systems
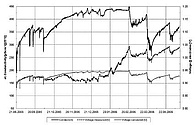
Calcium carbonate mineral formation constitutes a serious problem if deposition and clogging (scaling) impairs water transport in drainage systems. Some deposits are found to be porous and soft, while others are dense and hard. Obviously, the hard ones are more difficult to remove from a drainage system. Different types of plastic pipes of a drainage system may influence the nucleation and growth conditions and consistency of precipitated carbonates.
Our research focus on the nucleation and growth dynamics of carbonate minerals in relation to the different synthetic (plastic) materials. Special designed sinter alert sensors are used for in situ monitoring to track the ongoing scaling process remote-controlled.
Contact: Ronny Boch Michael Pettauer
Carbonates from Erzberg: Veins & iron ore Erzberg is well known as one of Austria’s most prominent places due to its historic, economic and scientific value. The pyramid shaped open pit iron ore mine hosts various carbonates including siderite, ankerite, dolomite, calcite and aragonite. The current research activities are focused on three major tasks:
- Evaluating the spatiotemporal growth dynamics and related environmental implications (chemical-sedimentary archive) from aragonite-calcite veins (“erzbergite”) in vertical fractures.
- Origin and radiometric age constraints of brittle fractures and faults based on CaCO3 precipitates.
- New insights to the formation conditions of the siderite-ankerite iron ore.
In this context, state-of-the-art solid- and fluid-phase geochemical and imaging techniques including various traditional and new stable isotope systems, multiply-substituted isotopologues (clumped isotopes), radiometric dating (238U-234U-230Th), elemental analysis (EPMA, LA-ICP-MS), high-resolution mineralogical/petrographic characterization, on-site aqueous solution monitoring and hydrogeochemical modeling are applied.
Contact: Ronny Boch
Scaling Forensics: Geothermal Energy Production Conditions Archived in Mineral Deposits
Scaling (unwanted precipitates) affecting different components (pipes, pumps, valves, heat exchangers) of geothermal facilities is a widespread obstacle during thermal water production from deep aquifers. The solid precipitates cause severe problems regarding geothermal energy extraction, i.e. clogging, reductions of inner diameters and energy transfers. Major preconditions are high dissolved solid and gas contents in combination with pressure and temperature changes. The scaling progress varies temporally and spatially depending on natural and man-made (operational) environmental conditions and the scale material itself constitutes a gradually developing mineral archive capturing variations of the critical production conditions in its composition and fabric. Applying high-resolution geochemical and petrographic analytical techniques the critical processes of scale formation are reconstructed, i.e. a forensic approach of these dead materials. Detailed solid-phase characterization is complemented by laboratory experiments and computer modelling.
A primary focus of our studies consists in a detailed analysis of the particular scale growth dynamics. The variable mineralization determines the overall progress (growth rate) and material properties (porosity, mechanical/chemical resistivity) of the scale deposits and consequently the necessity, timing and procedure of installation-specific maintenance intervals. In essence, our scaling forensic approach seeks for an increased process understanding of scaling (and related corrosion) with regard to unfavorable vs. favorable geothermal production conditions. A monitoring, optimization and preventive measures of installation-specific operational conditions are targeted.
Contact: Ronny Boch
Caves and Speleothems
Speleothems (dripstones) are used in paleoenvironmental research based on their potential to archive meteorological/climate information, their wide (global) distribution in karst areas, the high preservation potential underground and precise time (age) constraints based on uranium-series dating of these carbonates. Understanding the modern fluid-solid interactions is crucial for applying stalagmites of different time intervals as an archive of past environmental conditions.
Our research focuses on site-specific growth dynamics of modern (active) speleothems combining high-resolution petrographic, chemical and stable isotopic information captured in the actively growing stalagmites with increasingly automated (data loggers) field monitoring of drip water, cave air and CaCO3 precipitation on artificial substrates, and in comparison with data from regional meteorological stations. Katerloch Cave located north of Graz is well known for its overwhelming speleothem decoration and this cave was selected as an attractive study site.
Contact: Ronny Boch
Travertine & Carbonate Tufa: Formation & Paleoenvironment
Travertine deposits are typically laminated, consist of compact aragonite and/or calcite successions and are often associated with thermal waters, fault zones and microbial activity. Carbonate tufa, in contrast, is porous and consists of calcite precipitated from supersaturated stream waters of ambient atmospheric temperature. We study fossil and active sites of such freshwater carbonate formations focusing on the interaction of inorganic and organic processes and the (paleo)environmental information captured in their chemical composition.
The deposits can serve as indicators of past or active tectonic and mass movements and are further of value as a decoration- and building material. Two study sites are currently investigated: A major and compact Late Glacial travertine deposit in Eastern Tyrol and an active and vegetated stream precipitating carbonate tufa near Köttmannsdorf, Carinthia.
Contact: Ronny Boch