Lukas Roessler Escudero, Glen Smales, Viktor Hacker, Merit Bodner
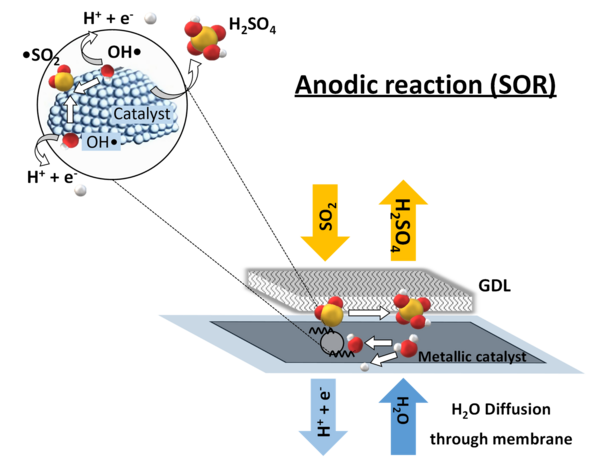
Sulphur depolarised electrolysis is an emerging electrochemical process with the potential to significantly reduce the energy demand of hydrogen production while enabling the co-production of valuable sulphuric acid. However, the technology is still in an early stage of development, with key operational parameters and catalyst configurations not yet fully optimised.
Guided by theoretical insights into the sulphur oxidation reaction, gold was identified as a highly promising anodic catalyst. This finding is particularly relevant in the context of reducing reliance on critical raw materials. The study combined in-situ electrochemical testing with advanced ex-situ characterisation techniques.
The optimisation of catalyst loading also yielded significant improvements. An asymmetric configuration (0.1 mgAu cm⁻²/0.3 mgPt cm⁻² for anode/cathode) achieved the best performance, reaching current densities above 0.65 A cm⁻² at 1.2 V. Compared to conventional Pt–Pt systems, this represents a substantial enhancement while simultaneously reducing platinum usage by 70% compared to the best literature reports and overall, 60% reduction in precious metal usage.
These results bring sulphur depolarised electrolysis closer to its performance targets and demonstrate viable pathways for improving efficiency and material sustainability. Marking an important step toward the competitive co-production of hydrogen and sulphuric acid within future integrated energy systems.
Journal of Cleaner Engineering and Technology
doi.org/10.1016/j.clet.2026.101201
Link to Article