Sebastian Sandner, Marija Zinaić, Marlene Kienberger
Purification of high-value biomolecules from biotech and biorefinery applications faces significant challenges since these products are produced in complex product streams and are inherently heat and pH sensitive. This makes their downstream processing prohibitively challenging and costly. Aqueous two-phase systems (ATPS) facilitate the process, as rapid phase separation allows high-volume processing, simplifying scale-up.
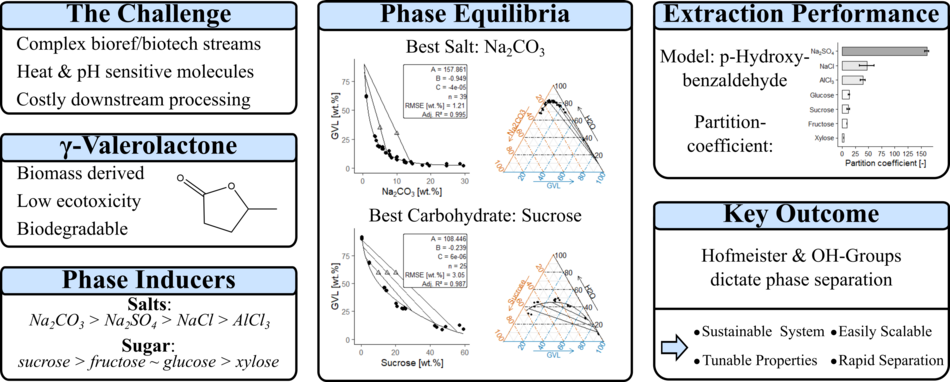
This work investigates aqueous two-phase systems involving γ-valerolactone (GVL)/water. GVL is a biomass derived, colorless cyclic ester, with low ecotoxicity and high biodegradability. The phase equilibria induced by four salts (NaCl, Na2SO4, Na2CO3, AlCl3) and four carbohydrates (xylose, glucose, fructose, sucrose) are described in XY and ternary diagrams. The salting out potential followed the order Na2CO3 > Na2SO4 > NaCl > AlCl3, which follows the order of the Hofmeister series. The sugaring-out potential followed the order sucrose > fructose ~ glucose > xylose, which correlated with the number of hydroxy groups. The ATPS were then applied to the liquid-liquid extraction of p-hydroxybenzaldehyde. At a mass ratio of 1:4:5 (salt:GVL:aqueous solution), and 2:4:4 (sugar:GVL:aqueous solution), p-hydroxybenzaldehyde partition coefficients of 161, 47, 39, were obtained with Na2SO4, NaCl, and AlCl3, respectively. Partition coefficients of 13, 12, 9, and 4 were obtained for glucose, sucrose, fructose, and xylose, respectively. The system containing Na2CO3 was not stable during liquid-liquid extraction.
This paper demonstrates the ability of salts and carbohydrates to induce phase separation of the GVL/water system. The gentle and tunable nature of these ATPS provides a highly versatile, non-denaturing platform for industrial purification of a diverse range of complex biomolecules.
Chemical Engineering Journal Advances
doi.org/10.1016/j.ceja.2026.101093
Link to Article